BY THE OPTIMIST DAILY EDITORIAL TEAM
Sexual health is getting a long-overdue upgrade. Thanks to recent approvals from the U.S. Food and Drug Administration, at-home STD testing is more accessible than ever, and it’s arriving alongside the first new gonorrhea treatments in decades. For public health experts, the timing couldn’t be better.
After years of climbing infection rates and pandemic-related disruptions to testing and treatment, these changes offer a hopeful shift. And while many challenges like high costs and public health funding cuts remain, the convenience and privacy of testing from home could help remove key barriers for people hesitant to get screened.
“Sexual health can be stigmatized and people can be hesitant about testing,” said Dr. Ina Park, a sexual health specialist at the University of California, San Francisco. “Now we have a lot of options for patients who may be wary of going into a provider’s office.”
Home testing is fast, private and accurate
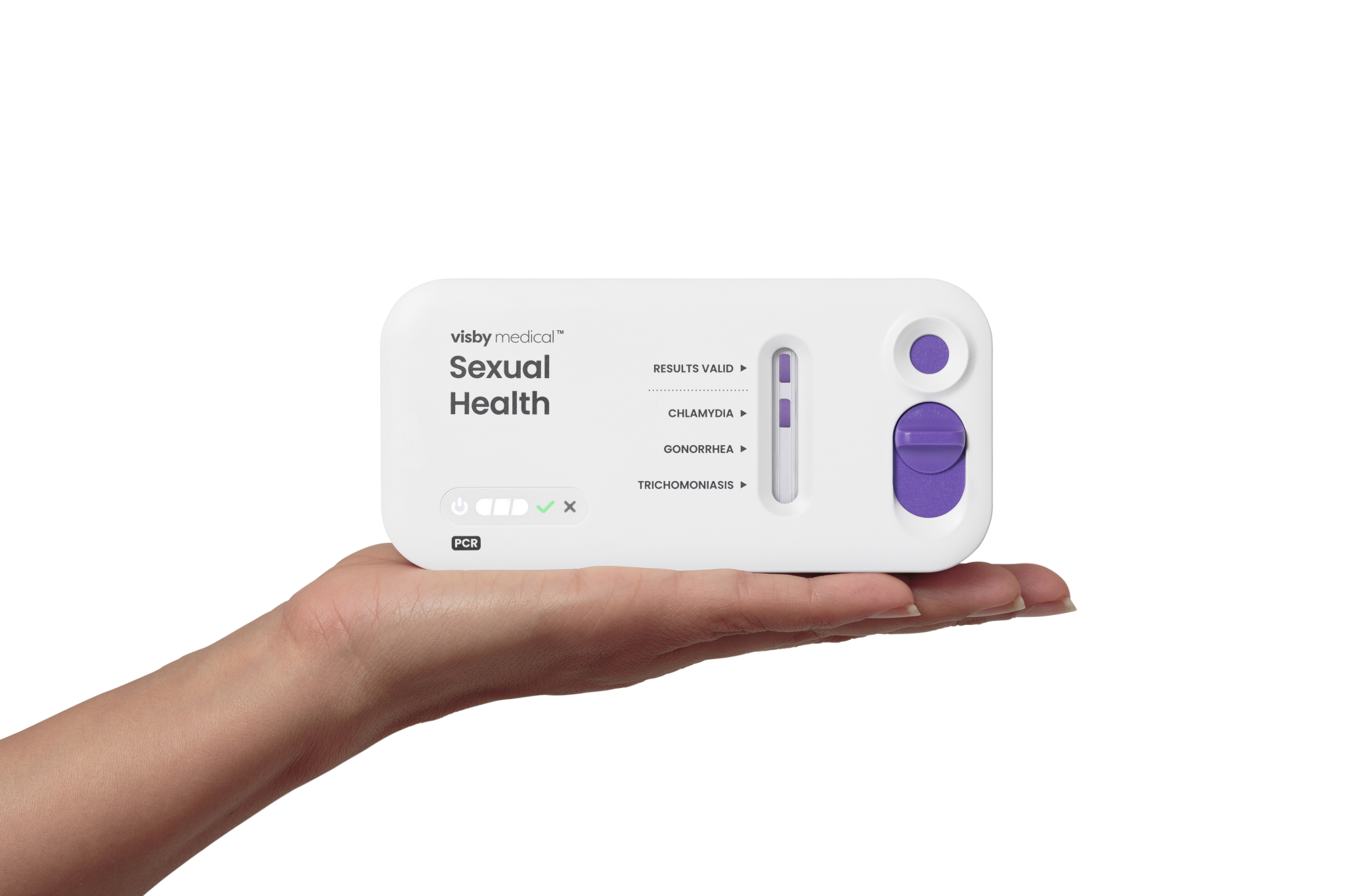
Last year, FDA approved Visby Medical’s all-in-one, at-home test that screens for gonorrhea, chlamydia, and trichomoniasis in women. The test includes a vaginal swab and a small diagnostic device. After collecting a sample, users upload a photo of the results to an app and can receive a telehealth consultation and prescription within as little as six hours.
Dr. Gary Schoolnik, Visby’s chief medical officer and a professor emeritus at Stanford, explained why that matters: “Many patients are very hard to track down and a lot of them, if they have a positive test result, are never treated and are lost to follow-up.” The streamlined model addresses this problem head-on.
The device was shown to detect the three infections with accuracy rates around 98 percent. That’s comparable to traditional lab tests, but it only costs $150. While not currently covered by insurance, it includes follow-up medical support and prescription access.
Other tests are designed to work with existing lab systems. Teal Health’s HPV self-collection kit, also approved by the FDA last year, allows users to collect a sample with the Teal Wand, ship it to a lab, and receive results from a provider. For the first time, federal guidelines now endorse self-collection for HPV screening—an important milestone in cervical cancer prevention.
New gonorrhea drugs offer promising alternatives
On the treatment side, another significant breakthrough has occurred; the FDA also approved two new oral medications for gonorrhea after years of limited options: Nuzolvenc and Bluejepa.
Both drugs can be taken by mouth, a notable improvement over the current go-to: an injection of ceftriaxone. The rise of drug-resistant gonorrhea has been a major concern, especially after the CDC dropped oral azithromycin from its guidelines due to resistance concerns.
“We were down to one class of antibiotics recommended to treat gonorrhea and we had no other good options,” Park said. “So to have two new options in the same year is very exciting.”
These drugs bring new hope in managing a notoriously adaptive infection that has become harder to treat over time.
The impact is already being felt
According to provisional data from the Centers for Disease Control and Prevention, STD rates are starting to decline. Gonorrhea cases have dropped for three consecutive years, and the most infectious forms of syphilis and adult cases of chlamydia have also seen a dip.
Experts point to multiple factors: less sexual activity among younger populations, a growing interest in “morning-after” antibiotics to prevent STDs, and wider access to at-home screening. While home testing is still relatively new, it’s beginning to reshape how people approach sexual health.
Still, experts caution that the shift to home testing could make tracking national infection rates more complicated. Many public health databases rely on centralized lab reporting, something not always captured when tests are done at home.
Another concern is cost and access. Tests like Visby’s $150 kit, while convenient, may be out of reach for lower-income individuals. And with recent funding cuts to the CDC and other public health agencies, there’s fear that the people who could benefit most from these innovations may be the last to access them.
“I’m feeling very optimistic about the fact that people have more testing options and also that we now have access to new drugs,” Park said. “What I fear is these cuts to public health are going to decrease access to sexual health care for populations who can least afford to take advantage of these new options.”
Did this solution stand out? Share it with a friend or support our mission by becoming an Emissary.