BY THE OPTIMIST DAILY EDITORIAL TEAM
For decades, the treatment for sleeping sickness was nearly as dangerous as the illness itself. One widely used intravenous drug caused a burning sensation in the veins and killed roughly one in 20 patients who received it. The oral replacement that followed required 10 days of pills and came with serious side effects, including vomiting and heart-rhythm disturbances severe enough to keep sick people from seeking care at all.
Then in March 2026, a committee of the European Medicines Agency cleared acoziborole: the first single-dose treatment for sleeping sickness. Three pills, swallowed together, once. The only side effect found in clinical trials was a mild to moderate headache.
“For decades, available treatments were difficult to use,” says Dr. Gerardo Priotto, who leads the World Health Organization’s efforts against sleeping sickness. “These challenges were especially severe in remote, rural areas, where most cases occur and health services are limited.”
How sleeping sickness works
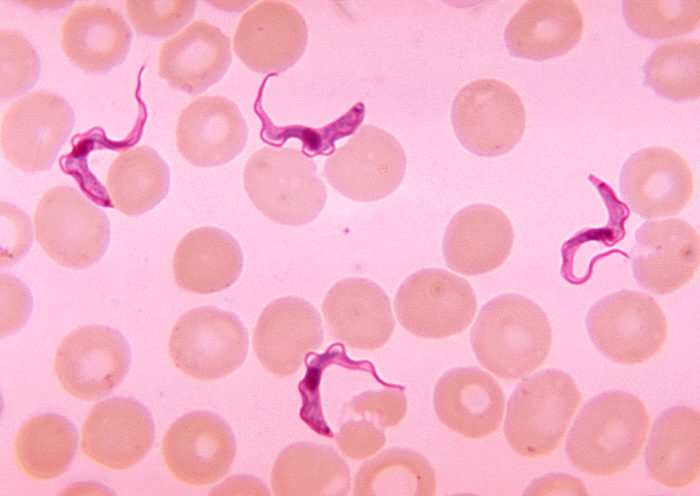
The illness spreads through the bite of a tsetse fly. The insect picks up the Trypanosoma brucei gambiense parasite from an infected person and passes it to the next person it bites. Tsetse flies thrive in warm savanna woodland and in vegetation along lakes and streams, which is why the illness shows up mostly in remote areas where people depend on fishing, hunting, and farming.
The illness moves in two stages. The first brings fever and headaches. The second, once the parasite crosses the blood-brain barrier, causes neurological effects: confusion, convulsions, and the disrupted sleep-wake cycles that the illness is named for. Patients grow drowsy during the day and wakeful at night. Without treatment, it progresses to coma and death. Some earlier drugs worked on one stage only. Acoziborole works on both.
From hundreds of thousands to one thousand
At its peak, sleeping sickness infected hundreds of thousands of people per year. Tsetse fly control programs, expanded diagnostic testing, and treatment access have driven that number down to roughly 1,000 cases annually, nearly two-thirds of them now in the Democratic Republic of Congo.
“Sleeping sickness has ebbed and returned at many points in history,” says Dr. Peter Hotez, dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston. The WHO’s goal is to eliminate the illness by 2030, and acoziborole is the drug health officials believe can get there.
“Its single-dose, well-tolerated regimen can dramatically simplify patient care, improve access to treatment and accelerate progress toward the elimination of sleeping sickness,” says Priotto. “It is a transformative tool for both patients and public health programs.”
The drug was developed by the Drugs for Neglected Diseases Initiative (DNDi), a nonprofit founded in 2003 around a hard reality: many illnesses in low-income countries were treatable, but no pharmaceutical company had a financial reason to develop those treatments. DNDi partnered with Sanofi on acoziborole, with funding from multiple sources including the Gates Foundation.
Trials built from scratch in the Congo
Dr. Wilfried Mutombo Kalonji, head of West and Central Africa Clinical Operations for DNDi, helped lead the clinical trials in South Ubangi, a remote province in the DRC. There was no infrastructure there to run a trial, so they built it. “Patients are often in remote areas, with no electricity, no water,” he says. “We had to set up all this, including training health workers, providing internet connections, electricity, and the safe way to get to testing sites, because they were remote. We overcame the challenges.”
His verdict: “We can’t dream to have better than this.”
Kalonji is now running a follow-up trial to see whether blood tests alone could allow treatment to begin the same day a patient is tested, skipping confirmatory tests that take longer, cost more, and can’t easily be done in the field. Monica Mugnier, who studies sleeping sickness at the Johns Hopkins Bloomberg School of Public Health, notes the parasite can sometimes evade detection, so a negative test doesn’t always mean a patient is clear. But if blood tests prove reliable enough, she says, “testing and treatment can begin on the same day,” a shift that could mean more patients treated and, potentially, the end of transmission once the human reservoir of the parasite is gone.
The funding question
Acoziborole still needs review by the DRC Ministry of Health and the WHO before treatment guidelines can be updated and other countries can authorize the drug. And global health experts are watching the funding picture.
“Like many tropical diseases, sleeping sickness is a disease of the poor,” says Hotez. Much of the progress so far has relied on international funding, and cuts to US and Western global health programs could limit the drug’s reach even after it clears every regulatory step.
“Many patients remember what the treatment was like for themselves or family members and were afraid of care,” says Dr. Stéphane Hugonnet, who worked on the clinical trials and leads DNDi’s sleeping sickness response. That fear kept people away even when they knew they were sick. Getting acoziborole to the communities that need it will require the same sustained commitment that brought case numbers down in the first place.
Did this solution stand out? Share it with a friend or support our mission by becoming an Emissary.